Everything we see — every colour, every shadow, every sunrise — begins with light. And light is one of the strangest things in the universe. It behaves as both a wave and a stream of particles at the same time. This is the wave-particle duality, the cornerstone of quantum physics. When light travels through space, it acts like a wave — oscillating electric and magnetic fields rippling outward. But when light hits a surface or is absorbed by your eye, it arrives in discrete packets of energy called photons. Each photon carries a specific amount of energy, and that energy determines the colour you see.
Think of it like the ocean. From a distance, the sea looks like continuous rolling waves. But zoom in close enough and you find that it is made of individual water molecules. Light works the same way — continuous from far away, granular up close. This was one of the great intellectual achievements of the 20th century: the realization that nature is not forced to be either a wave or a particle. It is both, depending on how you look at it.

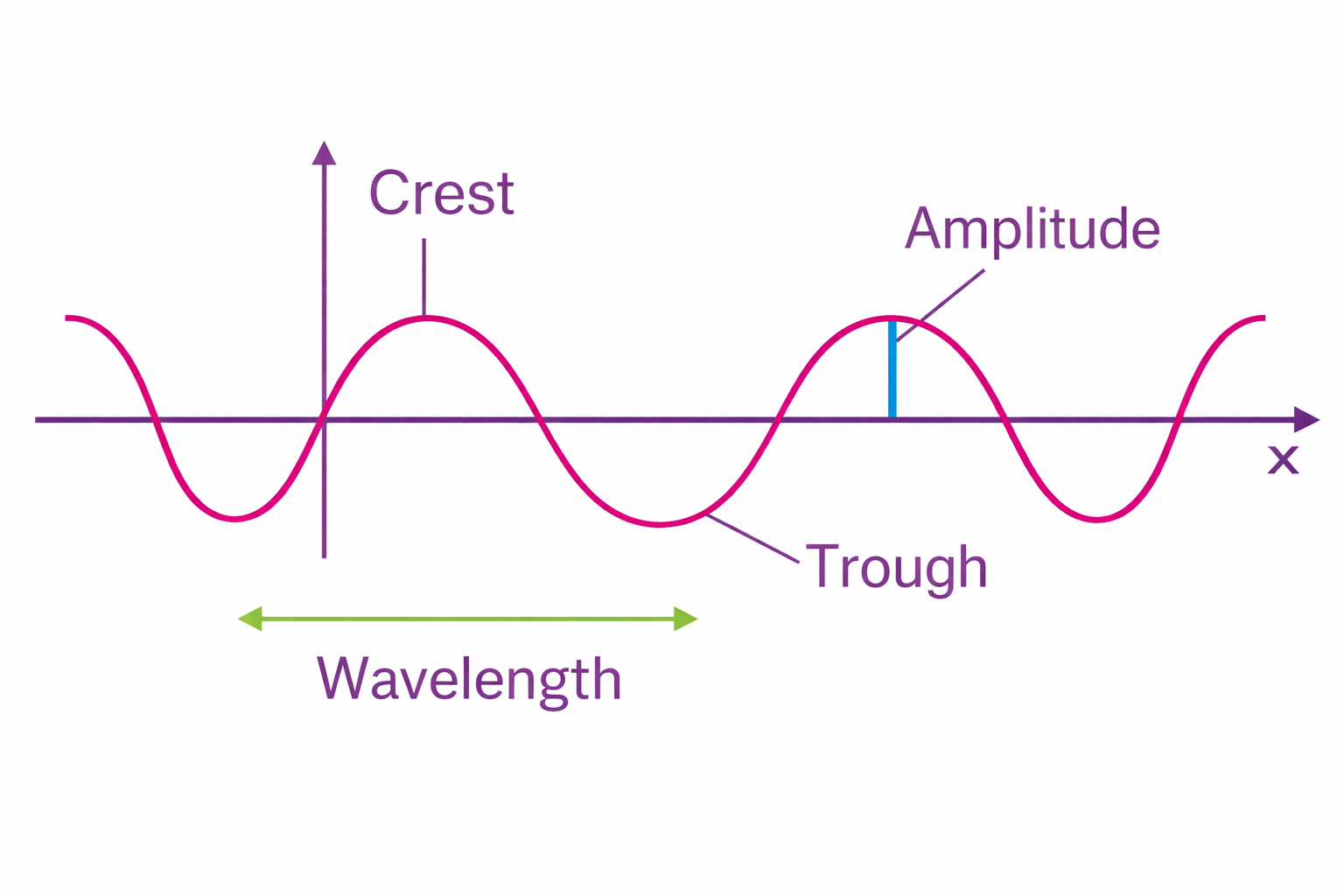
Light is part of the electromagnetic spectrum — the same family that includes radio waves, microwaves, X-rays, and gamma rays. What makes visible light special is simply that our eyes happen to detect it. The entire visible spectrum — everything from deep red to violet — occupies a tiny sliver of the electromagnetic spectrum, roughly between 380 and 700 nanometers in wavelength.
Wavelength and frequency are inversely related: the longer the wavelength, the lower the frequency, and the less energy the photon carries. Red light has the longest wavelength (~700 nm) and lowest energy. Violet light has the shortest (~380 nm) and highest energy. Everything in between — orange, yellow, green, blue — falls along this continuum. There is no sharp boundary between colours; the spectrum is a smooth gradient, and colour names are just labels we place upon it.
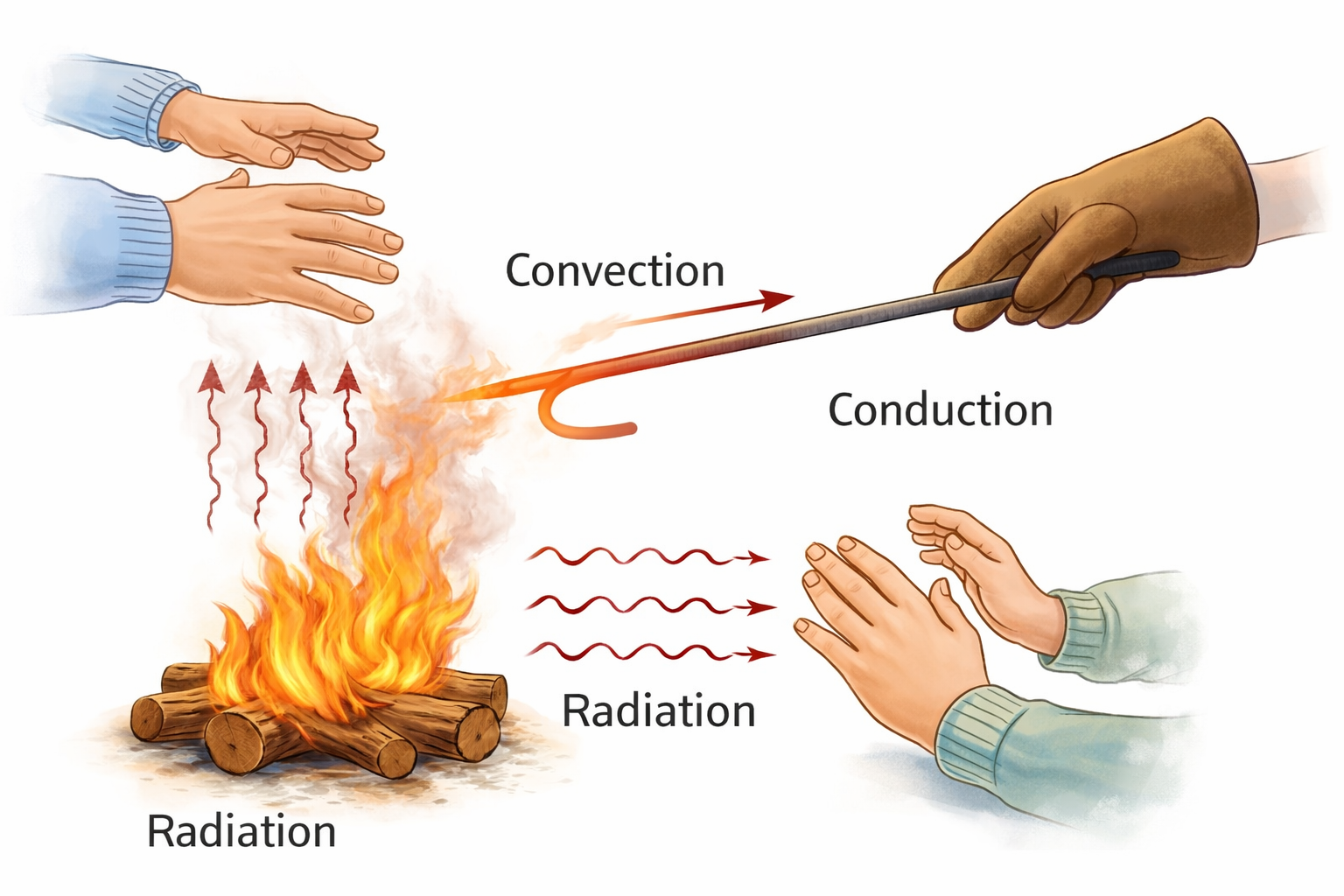
Every object in the universe with a temperature above absolute zero emits electromagnetic radiation. This is called thermal radiation, and it is a fundamental fact of physics. You emit it right now — in the infrared range, invisible to the naked eye but perfectly visible to a thermal camera.
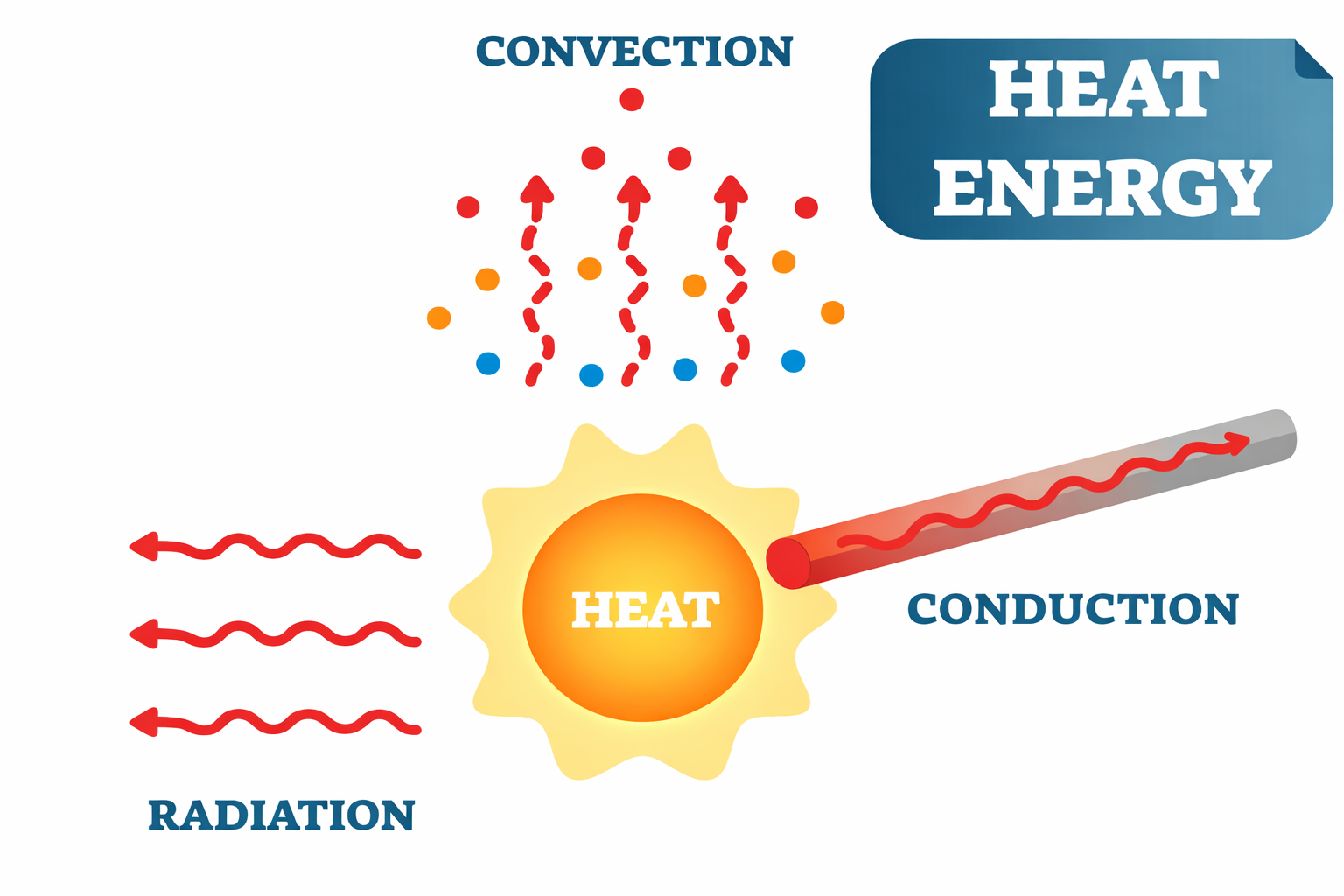
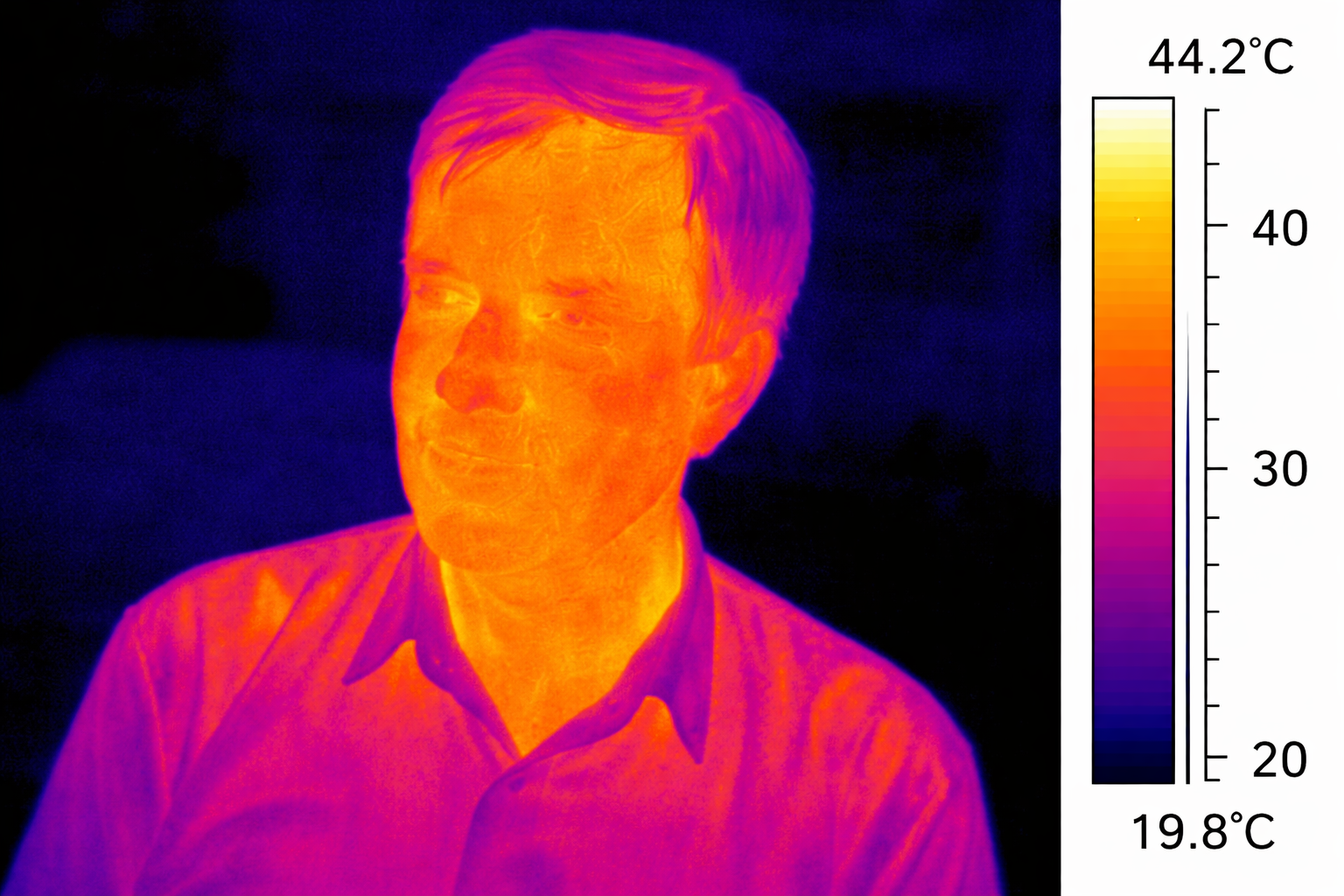
The hotter an object gets, the more energetic its radiation becomes, shifting to shorter wavelengths. Heat a piece of metal and it first glows dull red, then orange, then yellow, then white, and finally bluish-white. This progression is not random — it follows a precise relationship between temperature and the peak wavelength of emitted light. Physicists describe this using the concept of a black body — an idealised object that absorbs all radiation and re-emits it perfectly according to its temperature.
This is why we measure the “colour” of light sources in Kelvin:
Counter-intuitively, “warm” colours in lighting have lower colour temperatures, while “cool” bluish light has a higher temperature. A cosy candle at 1,800 K is physically cooler than the harsh blue sky at 10,000 K.
What makes stars so hot in the first place? The answer is thermonuclear fusion. Deep in a star’s core, under crushing pressure and temperatures exceeding 15 million °C, hydrogen nuclei fuse together to form helium. In the process, a tiny fraction of mass is converted into an enormous amount of energy — as described by Einstein’s famous E=mc². This is what powers a star for billions of years. As a star ages and exhausts its hydrogen, heavier stars can fuse helium into carbon, carbon into oxygen, and so on up the periodic table — all the way to iron, beyond which fusion no longer releases energy. At that point, massive stars collapse and explode as supernovae, scattering heavy elements into space — the very atoms that eventually form planets, oceans, and us.
Stars are natural black-body radiators, and their colour directly reveals their surface temperature. Red dwarf stars like Proxima Centauri burn at around 3,000 K — cool by stellar standards, glowing a deep orange-red. Our Sun, a yellow dwarf, has a surface temperature of about 5,778 K. Despite its name, when viewed from space (above the atmosphere), its light is actually white with a slight yellow tint.
Blue giant stars like Rigel in the constellation Orion burn at over 11,000 K, radiating intense bluish-white light. The hottest known stars, like the Wolf-Rayet stars, can exceed 200,000 K, emitting most of their energy in the ultraviolet range.


Why is the sky blue? The answer lies in a phenomenon called Rayleigh scattering. When sunlight enters the atmosphere, it collides with gas molecules (mainly nitrogen and oxygen). Shorter wavelengths — blue and violet — scatter much more strongly than longer wavelengths like red and yellow. The scattering intensity is inversely proportional to the fourth power of wavelength, meaning blue light scatters roughly 5.5 times more than red light.
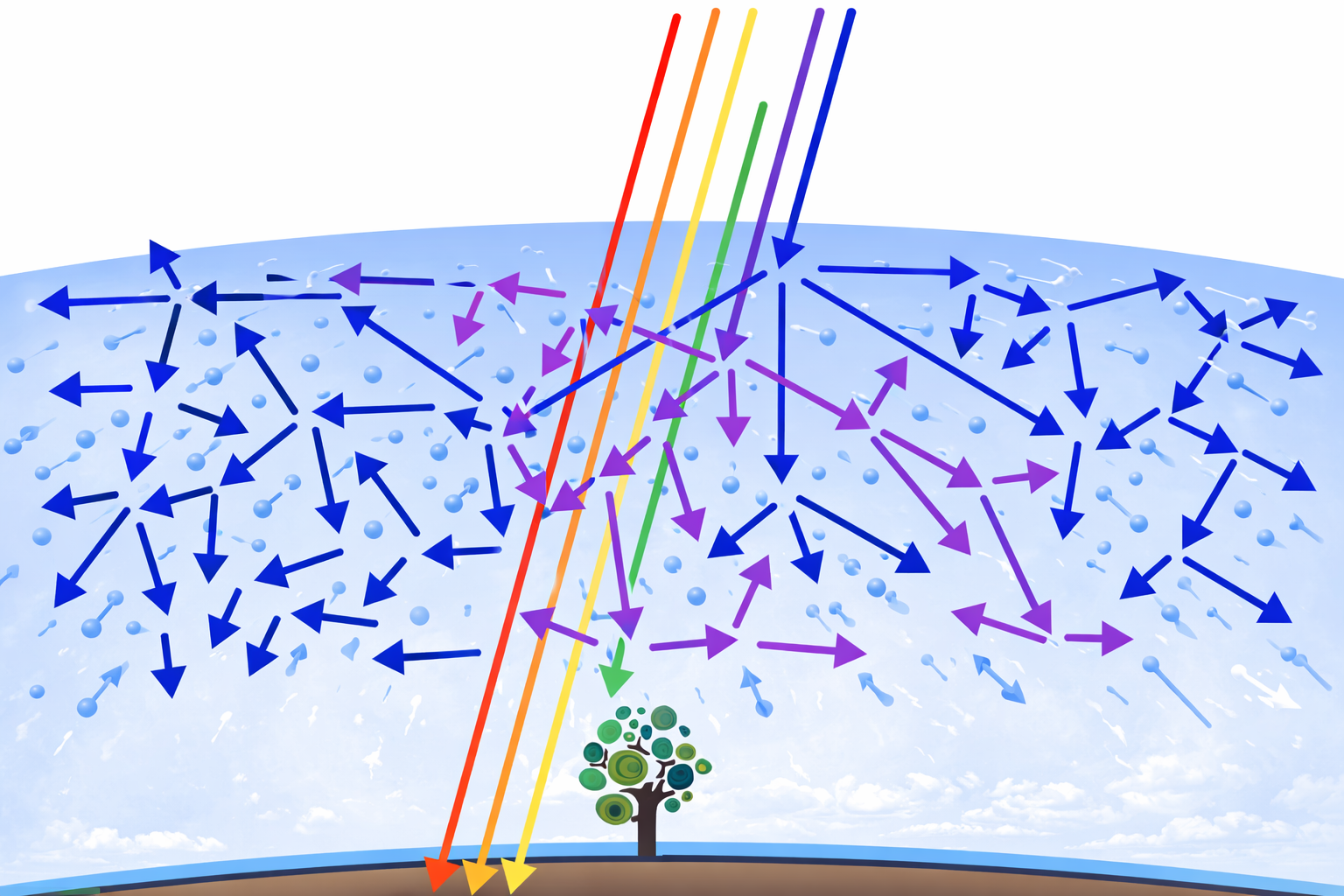
You might ask: if violet scatters even more than blue, why isn’t the sky violet? Two reasons. First, the Sun emits less violet light than blue. Second, our eyes are more sensitive to blue than to violet. The result: a vivid blue sky.
At sunset and sunrise, the Sun is near the horizon. Its light must travel through a much thicker layer of atmosphere to reach your eyes. By the time it arrives, most of the blue and green wavelengths have been scattered away, leaving primarily the long-wavelength red and orange light. The more atmosphere the light passes through, the redder it gets — which is why the Sun sometimes appears deep crimson just before it dips below the horizon.
When white light passes through a glass prism, it separates into a rainbow — a phenomenon called dispersion. This happens because different wavelengths of light travel at slightly different speeds through glass. Shorter wavelengths (violet, blue) slow down more and bend more sharply than longer wavelengths (red, orange). Isaac Newton was the first to demonstrate this systematically in the 1660s, proving that white light is not pure — it is a mixture of all visible colours.
Rainbows in the sky work on the same principle. Sunlight enters a raindrop, refracts (bends), reflects off the back of the drop, and refracts again on exit. Each wavelength bends slightly differently, spreading the light into the full spectrum. The reason a rainbow is an arc is a matter of geometry — each colour reaches your eye at a specific angle from the direction of the sunlight.

In 2014, Surrey NanoSystems created Vantablack — a material so dark that it absorbs 99.965% of visible light. The name stands for Vertically Aligned NanoTube Arrays. It is made of a forest of carbon nanotubes so densely packed that photons enter and bounce between the tubes until virtually all their energy is absorbed. Objects coated in Vantablack lose all visual depth and texture — they appear as flat silhouettes, as if a hole has been cut in reality. It approaches the theoretical ideal of a perfect black body absorber.
